Our general toxicology studies provide essential safety assessments for acute, subacute, and chronic exposure scenarios. These foundational studies help establish the safety profile of your compound and determine appropriate dosing parameters for further development. Our experienced team designs and executes comprehensive protocols that generate reliable data for regulatory submissions.
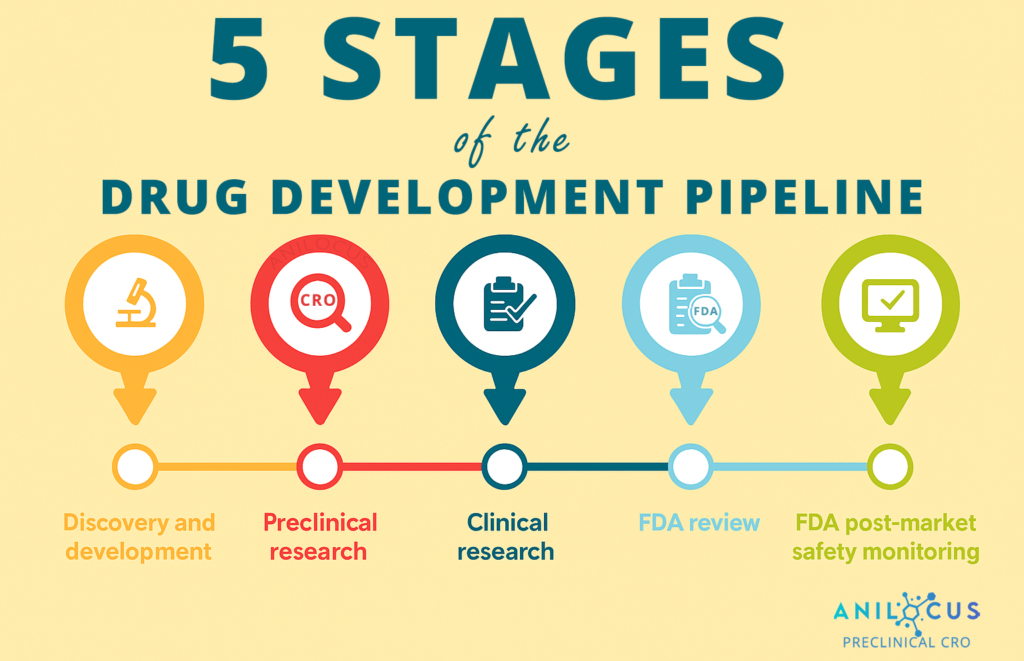
Supporting Your Development Pipeline
General toxicology studies form the backbone of preclinical safety assessment, providing critical data on how compounds affect living systems. These studies are essential for identifying potential adverse effects, establishing safe dose ranges, and understanding the overall risk-benefit profile of your drug candidate. From initial screening through regulatory submission, general toxicology data guides decision-making throughout the development process.
Successful general toxicology studies require careful protocol design, skilled technical execution, and thorough data interpretation. Our team combines extensive experience with state-of-the-art methodologies to deliver studies that meet regulatory standards while providing actionable insights for your development program.
Toxicology Services That Move Your Project Forward
Your drug development needs are unique and customized preclinical programs are the new normal. Our experts provide comprehensive general toxicology services designed to characterize the safety profile of your compound across multiple exposure scenarios.
Acute Toxicity Studies
- Single-dose toxicity assessment
- LD50 determination
- Rapid onset adverse effect identification
- Clinical observation scoring
- Mortality and morbidity monitoring
- Gross pathology evaluation
Subacute & Chronic Toxicity Studies
- 14-day to 90-day repeat-dose studies
- Chronic exposure assessment (6-month to 2-year)
- Dose-response relationship characterization
- Target organ identification
- Reversibility assessment
- Recovery period monitoring
Dose-Range Finding Studies
- Maximum Tolerated Dose (MTD) determination
- No Observed Adverse Effect Level (NOAEL) estimation
- Lowest Observed Adverse Effect Level (LOAEL) identification
- Dose escalation protocols
- Safety margin calculations
- Optimal dose selection for definitive studies
Clinical Observations & Monitoring
- Daily clinical observation scoring
- Body weight tracking
- Food and water consumption measurement
- Behavioral assessment
- Physical examination protocols
- Mortality and morbidity documentation
Organ Weight Analysis
- Terminal organ weight measurement
- Organ-to-body weight ratio calculations
- Organ-to-brain weight ratio analysis
- Statistical evaluation of organ weight changes
- Correlation with histopathological findings
Routes of Administration
- Oral (gavage, diet, and capsule)
- Parenteral (intravenous, subcutaneous, intramuscular, intraperitoneal)
- Dermal
- Inhalation
- Intranasal
- Ocular
- Infusion (bolus, intermittent, continuous)
Support Services
- Protocol design and regulatory guidance
- Dose formulation analysis
- Statistical analysis and reporting
- Toxicokinetic assessments
- Pathology services integration
- Regulatory submission support
- Study monitoring and quality assurance
Regulatory Compliance
Our general toxicology studies are designed to meet FDA, EMA, and other international regulatory requirements. We follow established guidelines including ICH, OECD, and EPA protocols to ensure your studies generate acceptable data for regulatory submissions worldwide.
Ready to advance your preclinical program?
Contact Anilocus today to discuss how our comprehensive general toxicology services can support your drug development goals. Talk to an Expert!



