Our genotoxicity studies assess the potential of test compounds to cause DNA damage or chromosomal aberrations. These critical safety assessments are required for regulatory approval and help identify mutagenic and carcinogenic risks early in development. Our comprehensive battery of genotoxicity assays provides the data needed to support your regulatory submissions and ensure patient safety.
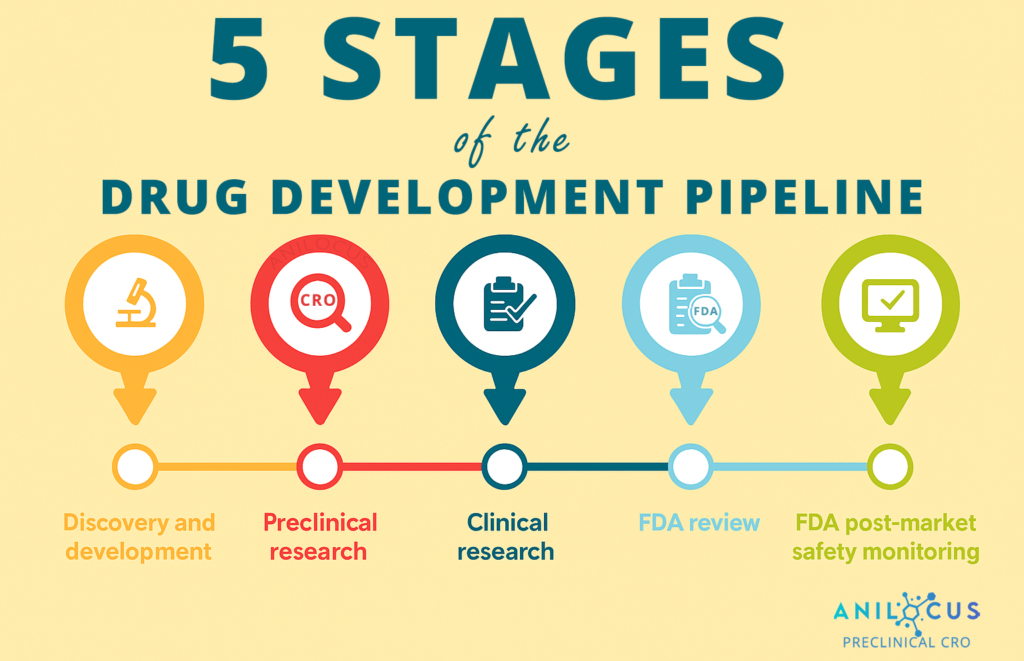
Supporting Your Development Pipeline
Genotoxicity testing is a fundamental component of preclinical research and safety assessments, required by regulatory authorities worldwide to evaluate the potential for genetic damage. These studies help identify compounds that may pose mutagenic or carcinogenic risks, enabling informed decision-making throughout the development process. Early genotoxicity screening can prevent costly late-stage failures and ensure patient safety.
Our genotoxicity testing follows internationally recognized guidelines and employs validated methodologies to deliver reliable, regulatory-compliant results. We offer both standard battery testing and specialized assays to meet the unique needs of your development program.
Toxicology Studies That Move Your Project Forward
Your drug development needs are unique and customized preclinical programs are the new normal. Our experts provide comprehensive genotoxicity testing services designed to identify potential genetic hazards and support regulatory submissions worldwide.
Bacterial Reverse Mutation Assay (Ames Test)
- Standard 5-strain bacterial test system
- Salmonella typhimurium strains (TA98, TA100, TA1535, TA1537)
- Escherichia coli strain (WP2 uvrA)
- Metabolic activation with S9 fraction
- Dose-response evaluation
- Positive and negative control validation
In Vitro Micronucleus Assay
- Mammalian cell line testing (CHO, CHL, HepG2)
- Chromosomal damage assessment
- Automated scoring systems available
- Metabolic activation studies
- Cytotoxicity evaluation
- Statistical analysis and interpretation
Chromosomal Aberration Assay
- Structural chromosome damage detection
- Mammalian cell culture systems
- Metaphase chromosome analysis
- Gap and break scoring
- Chromatid and chromosome aberration classification
- Mitotic index determination
Comet Assay (Single Cell Gel Electrophoresis)
- DNA strand break detection
- Primary cell and cell line compatibility
- Alkaline and neutral comet protocols
- Automated image analysis
- Oxidative DNA damage assessment
- Repair kinetics evaluation
Mouse Lymphoma TK Assay
- Gene mutation detection
- L5178Y mouse lymphoma cells
- Forward mutation analysis
- Small and large colony phenotyping
- Metabolic activation studies
- Clastogenic potential assessment
Specialized Genotoxicity Assays
- In vivo micronucleus assay
- Transgenic rodent gene mutation assays
- DNA adduct analysis
- Unscheduled DNA synthesis (UDS) assay
- Sister chromatid exchange (SCE) assay
- Mammalian cell transformation assay
Regulatory Compliance
Our genotoxicity studies are designed to meet FDA, EMA, and other international regulatory requirements. We follow ICH S2(R1) guidelines and OECD test protocols to ensure your studies generate acceptable data for regulatory submissions worldwide.
Ready to advance your preclinical program?
Contact Anilocus today to discuss how our comprehensive genotoxicity services can support your drug development goals. Talk to an Expert!



