Our toxicology studies are powered by multidisciplinary scientists and study directors who conduct comprehensive preclinical programs. When developing therapeutics, agrochemicals, chemicals, or medical devices, you can trust that your in vitro and in vivo toxicology studies will be designed, executed, and delivered in a way that best characterizes the potential for human toxicity.
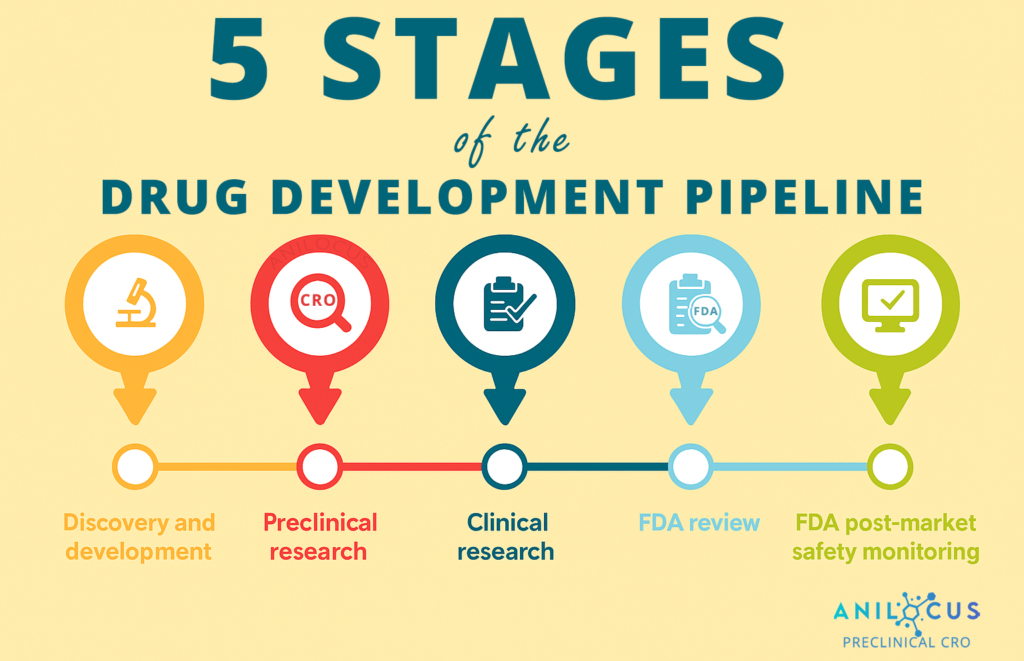
Supporting Your Development Pipeline
Preclinical toxicology studies are an essential part of drug development as they help to evaluate the potential safety and toxicity of a drug candidate before it is tested in human clinical trials. These studies play a pivotal role in identifying the appropriate dose range for a drug candidate and can provide information about potential side effects and adverse reactions. Preclinical toxicology studies help determine the potential risks and benefits of a drug candidate and can provide a basis for regulatory approval.
Successful toxicology studies demand the contributions of skilled study directors, toxicologists, pathologists, veterinarians, surgeons, regulatory specialists, and support personnel. With a strong mechanistic understanding of toxicology, our team can efficiently determine potential risk factors early in the drug development process. Many of our scientists have served on, or are currently participating in, regulatory and industry body working groups, shaping the future of our studies.
Toxicology Services That Move Your Project Forward
Your drug development needs are unique and customized preclinical programs are the new normal. Our experts have more than just general toxicology knowledge – explore all the specialty toxicology services available to you.
General Toxicology Studies
- Maximum Tolerated Dose (MTD) determination
- No Observed Adverse Effect Level (NOAEL) estimation
- LD50 determination
- Dose-range finding studies
- Body weight tracking and clinical observations
- Food/water consumption measurement
- Organ weight measurement
Clinical Pathology Services
Comprehensive blood, serum, and urine analysis including:
Hematology
- Complete blood counts with differential
- RBC, WBC, hemoglobin, hematocrit
- Platelet count and red blood cell indices
Clinical Chemistry
- Liver function markers (ALT, AST, ALP)
- Kidney function parameters (Creatinine, BUN)
- Metabolic indicators (Glucose, Albumin, Total protein)
- Comprehensive electrolyte panels
Urinalysis
- pH, specific gravity, protein, glucose
- Ketone detection and microscopic sediment analysis
Pathology Services
- Full necropsy procedures
- Organ collection and organ-to-body weight ratios
- Tissue fixation and histological processing
- Hematoxylin & eosin (H&E) staining
- Microscopic examination with lesion scoring
- Optional special stains (PAS, Trichrome)
Genotoxicity Studies
- Ames test (Bacterial Reverse Mutation Assay)
- In vitro micronucleus assay
- Chromosomal aberration assay
- Comet assay (Single Cell Gel Electrophoresis)
- Mouse lymphoma TK assay
Reproductive & Developmental Toxicology
- Sperm morphology and motility assessment
- Estrous cycle tracking
- Fertility index calculation
- Embryo-fetal development studies
- Teratogenicity scoring (skeletal and visceral exams)
- Prenatal/postnatal development assessment
Immunotoxicity Assessment
- T-cell proliferation assay
- Natural killer (NK) cell cytotoxicity testing
- Cytokine profiling (IL-2, IFN-γ)
- Delayed-type hypersensitivity (DTH) assay
- Flow cytometry for immune cell subsets
Safety Pharmacology-Linked Studies
- hERG channel inhibition assay (in vitro)
- ECG telemetry in rodents and non-human primates
- Respiratory monitoring (whole-body plethysmography)
- Locomotor activity and rotarod testing
- Core body temperature measurement
Carcinogenicity Assessment
- Cell transformation assays
- DNA adduct detection
- Initiation-promotion studies
- Transgenic mouse assays (rasH2 model)
- 2-year rodent bioassays
Routes of Administration
- Dermal
- Inhalation
- Intranasal
- Intrathecal
- Ocular
- Oral (gavage, diet, and capsule)
- Parenteral (intravenous, subcutaneous, intradermal, intramuscular, intraperitoneal)
- Infusion (bolus, intermittent, continuous)
Support Services
- Analytical chemistry for dose formulation analysis
- Bioanalytical chemistry sample assays for drug concentration
- Immunology endpoint assessments
- Pathology services
- Scientific and strategic consulting
- Specialized statistical analyses
- Toxicokinetics and pharmacokinetics
Regulatory Compliance
Our toxicology services are designed to meet FDA, EMA, and other international regulatory requirements. Our experienced team works closely with sponsors to design appropriate study protocols that generate robust data for regulatory submissions.
Ready to advance your preclinical program?
Contact Anilocus today to discuss how our comprehensive toxicology services can support your drug development goals. Talk to an Expert!



