Our mutagenicity studies evaluate the potential of test compounds to induce gene mutations in bacterial and mammalian systems. These essential safety assessments detect point mutations, base-pair substitutions, and frameshift mutations that could lead to carcinogenic potential. Our comprehensive mutagenicity testing battery provides critical data for regulatory submissions and risk assessment.
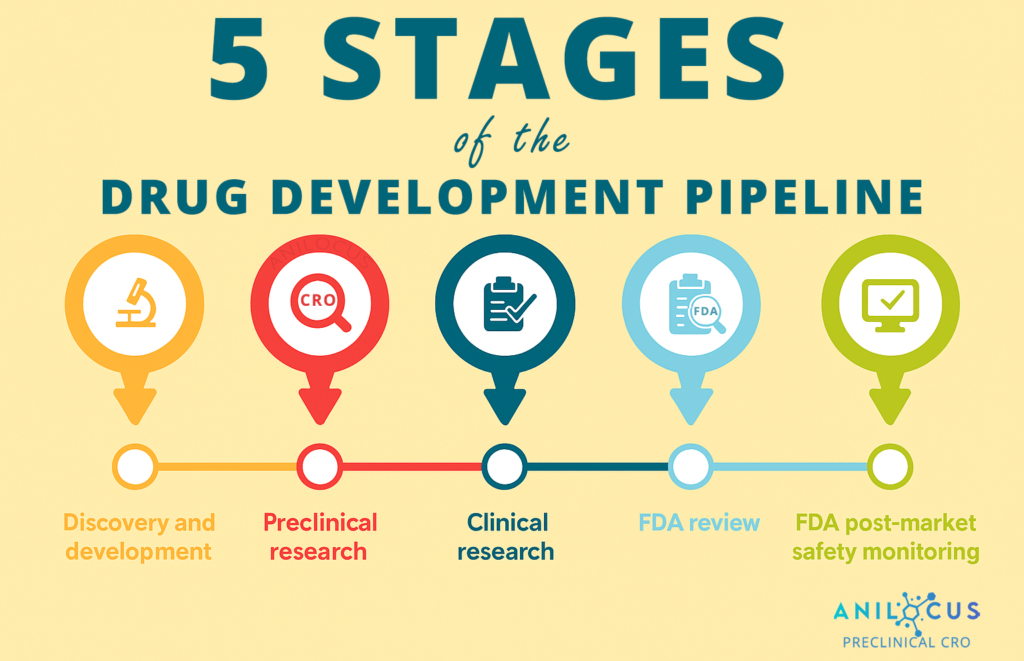
Supporting Your Development Pipeline
Mutagenicity testing is a cornerstone of preclinical research and safety assessments, providing essential information about a compound’s potential to cause genetic mutations. These studies are required by regulatory authorities worldwide and serve as key indicators of carcinogenic potential. Early mutagenicity screening helps identify problematic compounds before significant resources are invested, while negative results support the safety profile of promising drug candidates.
Our mutagenicity testing employs validated methodologies and follows internationally recognized guidelines to deliver reliable, regulatory-compliant results. We provide both standard screening assays and specialized mutation studies to comprehensively evaluate your compound’s mutagenic potential.
Toxicology Studies That Move Your Project Forward
Your drug development needs are unique and customized preclinical programs are the new normal. Our experts provide comprehensive mutagenicity testing services designed to detect gene mutations and support regulatory submissions with confidence.
Bacterial Mutation Assays
- Salmonella typhimurium reverse mutation assay (Ames test)
- Multiple strain testing (TA98, TA100, TA1535, TA1537, TA102)
- Escherichia coli WP2 uvrA strain testing
- Base-pair substitution detection
- Frameshift mutation identification
- Metabolic activation with rat liver S9
Mammalian Cell Gene Mutation Assays
- Mouse lymphoma L5178Y TK+/- assay
- CHO/HPRT gene mutation assay
- V79/HPRT gene mutation assay
- Forward mutation detection
- Colony sizing and phenotype analysis
- Cloning efficiency determination
In Vivo Mutation Assays
- Transgenic rodent mutation assays
- Big Blue® lacI transgenic mouse model
- MutaMouse® lacZ transgenic model
- Tissue-specific mutation analysis
- Multiple organ mutation screening
- Molecular mutation characterization
Specialized Mutation Detection
- Pig-a gene mutation assay
- Duplex sequencing for rare mutations
- Site-specific mutation analysis
- Mutation spectrum characterization
- DNA repair deficiency screening
- Hypoxanthine-guanine phosphoribosyltransferase (HPRT) assays
Mutation Mechanism Studies
- Base excision repair pathway analysis
- Nucleotide excision repair assessment
- Mismatch repair evaluation
- DNA polymerase fidelity testing
- Oxidative damage mutation patterns
- Alkylation-induced mutation profiling
Advanced Mutation Analysis
- Next-generation sequencing mutation detection
- Quantitative PCR mutation analysis
- Mutation frequency calculations
- Statistical mutation rate analysis
- Dose-response mutation modeling
- Comparative mutagenicity assessment
Regulatory Compliance
Our mutagenicity studies are designed to meet FDA, EMA, and other international regulatory requirements. We follow ICH S2(R1) guidelines, OECD test protocols, and EPA testing standards to ensure your studies generate acceptable data for regulatory submissions worldwide.
Ready to advance your preclinical program?
Contact Anilocus today to discuss how our comprehensive mutagenicity services can support your drug development goals. Talk to an Expert!



